



Our group has a range of research interests that lie on the chemical biology interface, from developing novel chemical small molecule probes, inhibitors and delivery vehicles to the discovery and application of new synthetic methodology for their construction. All this provides us with the tools to challenge and answer important biological and chemical questions.
Coordination polymer networks (CPs) and metal–organic frameworks (MOFs) were originally sought for their structural stability and rigidity. However, more recently the focus has been to engineer dynamic ‘sponge-like’, porous, frameworks that offer greater opportunity for unique structural transformations. These dynamic frameworks allow transformations that lead to interesting and unique applications, such as enhanced proton conductivity, plastically flexible crystals, controlled drug delivery, increased gas storage capabilities and uses as mechanical shock absorbers. Several external stimuli, such as light, heat and pressure can be utilised to create conditions to manipulate the dynamic nature of these frameworks.
Flexible coordination polymer networks reveal a plethora of structural transformations when exposed to high pressure as an external stimulus. Transformations are driven through organic linkers in a variety of ways, such as bond rotations, rearrangements and conformational shifts. We have successfully synthesised two sets of isostructural lanthanide coordination polymer networks that differ as a consequence of the lanthanide contraction. We have demonstrated that a pressure-induced reversible phase transition is possible between the two structurally distinct sets of networks. The system demonstrates a bond rearrangement due to an alteration of carboxylic acid binding modes from the 2,5-bis(allyloxy)terephthalate linker in addition to bond rotations within the flexible carbon backbone of the linker between 30–35 kbar.
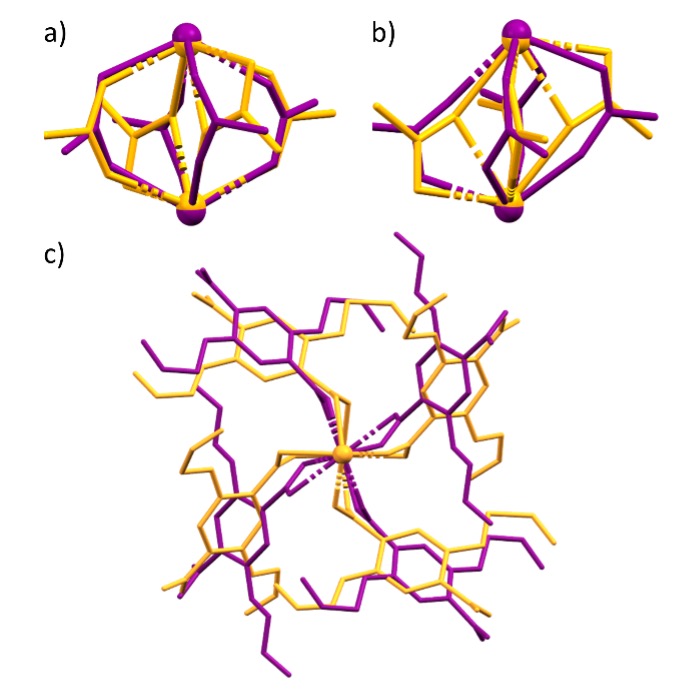
Figure 1: Overlaid views of Pr complex at atmospheric pressure (orange) and at 35 kbar (purple). a) and b) Show two different orientations of the overlaid secondary building units, whereby you can visualise the change in carboxylic acid binding mode. c.) Shows a view along eclipsing metal centres, where you can see the anti-clockwise rotation of linkers under high pressure.
This project investigates the potential use of (Z)-stilbenedicarboxylic acid as an organic linker for the fabrication of MOFs. This linker is selected for its alkene functionality despite being previously overlooked when compared to its E-isomer counterpart. Alkene functionalities provide the ability to undergo photo cyclisation when exposed to a UV-light stimulus, thus when incorporated into a framework, potentially creating 3rd generation dynamic MOFs. This project involves the synthesis of novel lanthanide-based frameworks and subsequent crystal structure analysis and characterisation. Two subsets of frameworks are isolated using the same crystallisation conditions, with one subset exhibiting a new underlying net topology that has been analysed in detail. Furthermore, the luminescent properties of the generated lanthanide frameworks are explored to expand our understanding of these materials.

Images of Eu stilbene crystals taken at varying magnifications (a) x1 b) x4 c) x2 d) x4), showing crystal quality varies within sample batches
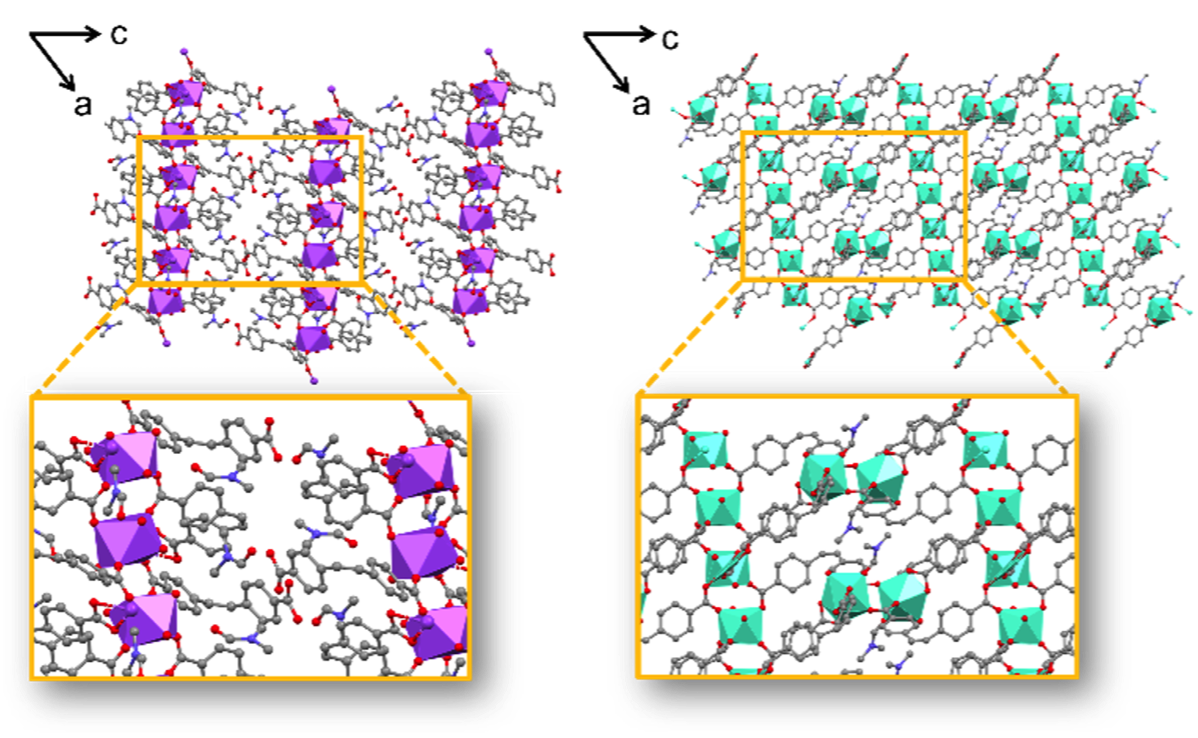
Comparison of 2D and 3D crystal structures of Eu2((Z)-SDC)3(H2O)2(DMF) viewed down the [010] crystallographic axis

Biosciences Institute
Faculty of Medical Sciences
Newcastle University
Cookson Building
Newcastle-upon-Tyne
NE2 4HH, UK
Tel.: +44 (191) 2082357
Email: jon.sellars@newcastle.ac.uk
///blast.palace.cycles